Value it Brings to the Outcome of a Trial
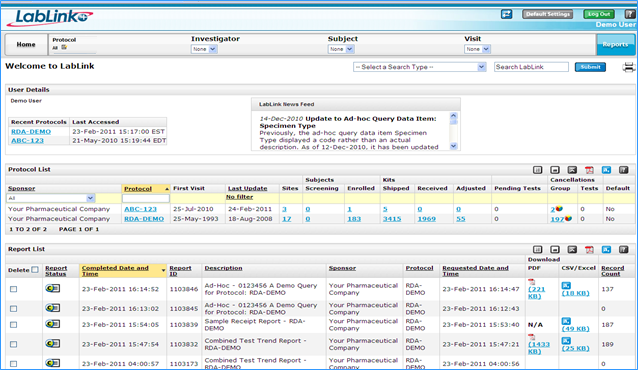
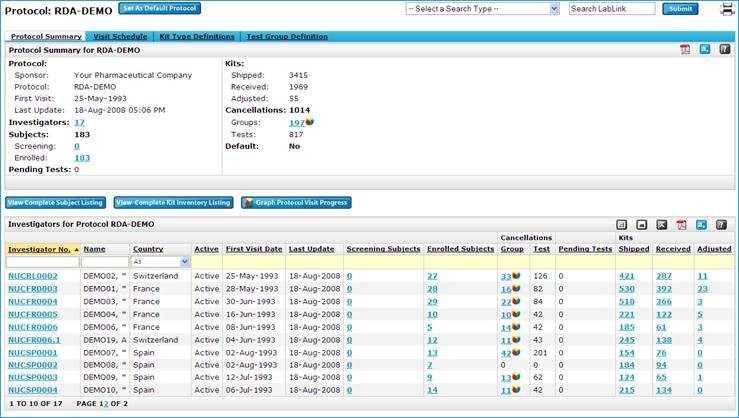
In order to be assured of the quality and integrity of lab test data, LabLink+ provides sponsors with 24-hour monitoring of kit locations. Additionally, Labcorp LabLink+ has the enhanced ability to generate and schedule customized reports, which saves time and increases the efficiency and consistency of study teams.